2016 Vol.28(5)
Display Mode: |
2016, 28(5): 467-477.
doi: 10.21147/j.issn.1000-9604.2016.05.01
Abstract:
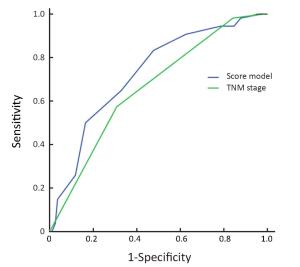
Objective In our previous work, we incorporated complete blood count (CBC) into TNM stage to develop a new prognostic score model, which was validated to improve prediction efficiency of TNM stage for nasopharyngeal carcinoma (NPC). The purpose of this study was to revalidate the accuracy of the model, and its superiority to TNM stage, through data from a prospective study. Methods CBC of 249 eligible patients from the 863 Program No. 2006AA02Z4B4 was evaluated. Prognostic index (PI) of each patient was calculated according to the score model. Then they were divided by the PI into three categories: the low-, intermediate-and high-risk patients. The 5-year disease-specific survival (DSS) of the three categories was compared by a log-rank test. The model and TNM stage (7th edition) were compared on efficiency for predicting the 5-year DSS, through comparison of the area under curve (AUC) of their receiver-operating characteristic curves. Results The 5-year DSS of the low-, intermediate-and high-risk patients were 96.0%, 79.1% and 62.2%, respectively. The low-and intermediate-risk patients had better DSS than the high-risk patients (P<0.001 and P<0.005, respectively). And there was a trend of better DSS in the low-risk patients, compared with the intermediate-risk patients (P=0.049). The AUC of the model was larger than that of TNM stage (0.726 vs. 0.661, P=0.023). Conclusions A CBC-based prognostic score model was revalidated to be accurate and superior to TNM stage on predicting 5-year DSS of NPC.
2016, 28(5): 478-485.
doi: 10.21147/j.issn.1000-9604.2016.05.02
Abstract:
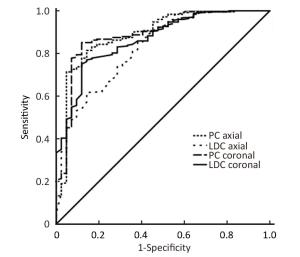
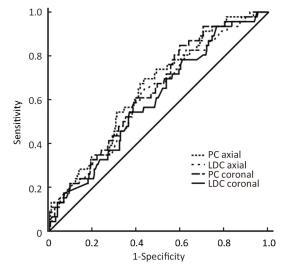
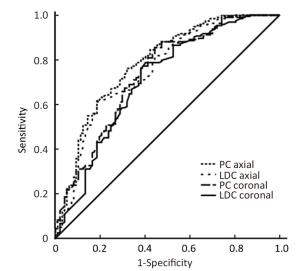
Objective Early assessment of response to neoadjuvant chemotherapy (NAC) for breast cancer allows therapy to be individualized. The optimal assessment method has not been established. We investigated the accuracy of automated breast ultrasound (ABUS) to predict pathological outcomes after NAC. Methods A total of 290 breast cancer patients were eligible for this study. Tumor response after 2 cycles of chemotherapy was assessed using the product change of two largest perpendicular diameters (PC) or the longest diameter change (LDC). PC and LDC were analyzed on the axial and the coronal planes respectively. Receiver operating characteristic (ROC) curves were used to evaluate overall performance of the prediction methods. Youden's indexes were calculated to select the optimal cut-off value for each method. Sensitivity, specificity, positive and negative predictive values (PPV and NPV) and the area under the ROC curve (AUC) were calculated accordingly. Results ypT0/is was achieved in 42 patients (14.5%) while ypT0 was achieved in 30 patients (10.3%) after NAC. All four prediction methods (PC on axial planes, LDC on axial planes, PC on coronal planes and LDC on coronal planes) displayed high AUCs (all>0.82), with the highest of 0.89 [95% confidence interval (95% CI), 0.83-0.95] when mid-treatment ABUS was used to predict final pathological complete remission (pCR). High sensitivities (85.7%-88.1%) were observed across all four prediction methods while high specificities (81.5%-85.1%) were observed in two methods used PC. The optimal cut-off values defined by our data replicate the WHO and the RECIST criteria. Lower AUCs were observed when mid-treatment ABUS was used to predict poor pathological outcomes. Conclusions ABUS is a useful tool in early evaluation of pCR after NAC while less reliable when predicting poor pathological outcomes.
2016, 28(5): 486-494.
doi: 10.21147/j.issn.1000-9604.2016.05.03
Abstract:
Objective Combined small cell lung cancer (C-SCLC) is an uncommon subgroup of small cell lung cancer (SCLC) and few clinical data can be referred. Our study is to investigate the clinical features and prognostic factors of C-SCLC, as well as the role of multimodality treatment. Methods Between January 2004 and December 2012, patients with histologically diagnosed C-SCLC were retrospectively analyzed. The survivals were evaluated with the Kaplan-Meier method. Univariate and multivariate analyses were used to evaluate potential prognostic factors. Results One hundred and fourteen patients were enrolled, with a median age of 59 (range: 20−79) years old. The most common combined component was squamous cell carcinoma (52.6%). Among these patients, the disease was stage I, II, III and IV in 9.6%, 19.3%, 46.5% and 24.6% of the patients, respectively. Eighty patients (70.2%) received at least two of the three modalities containing chemotherapy, radiotherapy and surgery. The median follow-up was 32.5 months. The median time of overall survival (OS) was 26.2 months. On univariate analysis, smoking (P=0.029), Karnofsky performance score (KPS) <80 (P=0.000), advanced TNM stage (P=0.000), no surgery (P=0.010), positive resection margin (P=0.000), positive lymph nodes ≥4 (P=0.000), positive lymph node ratio >10% (P=0.000) and non-multimodality treatment (P=0.004) were associated with poor OS. Multivariate analysis confirmed that smoking, advanced TNM stage, positive resection margin and positive lymph nodes ratio >10% were poor prognostic features. Conclusions C-SCLC has a relatively early stage and good prognosis, which may due to the underestimated diagnosis in non-surgical patients. Multimodality therapy is recommended, especially for limited disease. Smoking, advanced TNM stage, positive resection margin and positive lymph nodes ratio >10% are poor prognostic factors.
2016, 28(5): 495-502.
doi: 10.21147/j.issn.1000-9604.2016.05.04
Abstract:
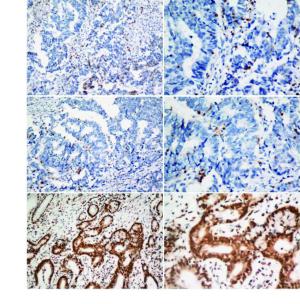
Objective The novel fully automated immunohistochemistry (IHC) assay-Ventana anaplastic lymphoma kinase (ALK)-D5F3 for screening ALK rearrangements has been approved by China’s Food and Drug Administration in 2013, our previous study disclosed a highly specificity and sensitivity nearly 100%, and its efficacy needs to be evaluated in a large cohort of primary lung adenocarcinoma patients, and to compare clinicopathological features with ALK (+) and ALK (?) lung adenocarcinoma. Methods A total of 1, 504 consecutive surgical lung adenocarcinoma cases of Chinese Han population were collected and re-diagnosed according to the 2011 multidisciplinary classification of lung adenocarcinoma. Fully automated Ventana ALK-D5F3 IHC staining with a binary scoring was adopted to evaluate staining and correlated with clinicopathological characters, including age, sex, differentiation degree, histological subtype, lymph node metastasis, and clinical staging. ALK (+) patients were followed-up, and targeted therapy of ALK-inhibitors was adopted and observed in patients with stage IV according to the NCCN guideline. Results ALK positive adenocarcinomas were identified in 6.6% of the surgically resected 1, 504 NSCLCs, and significantly younger than the negative group (P<0.05).Mucinous adenocarcinoma (28.2%) was determined to be predominant in ALK (+) cases, followed by the solid type (11.7%), specific type (6.8%), papillary type (5.6%), acinar type (5.5%), and lepidic type (3.1%), and the differences were statistically significant (χ2=42.011, P<0.05). ALK (+) adenocarcinoma with lymph node metastasis (10.8%) were significantly higher than that without lymph node metastasis (4.5%) (χ2=19.809, P<0.05); and ALK (+) in phase IV (20%) was significantly higher than phase III (12.9%), phase II (4.2%), phase I (4.5%), and phase 0 (0) (χ2=36.068, P<0.05). Multivariate logistic regression disclosed that patient age, AJCC staging, and histological mucinous subtype were correlated with ALK positive staining (OR=0.959, 1.578, 5.036, respectively). Sixty eight patients had followed-up results, five patients out of which primarily diagnosed or progressed into Stage IV benefited well from targeted therapy with Crizotinib. Conclusions The ALK fusion protein was seen in 6.6% Chinese NSCLC patients, and mostly seen in younger, clinically higher staging, mucinous and solid predominant adenocarcinoma. Clinical trials in patients of Stage IV confirmed that ALK-D5F3 Ventana IHC is serviceable in screening ALK-positive candidates for molecular targeted therapy.
2016, 28(5): 503-510.
doi: 10.21147/j.issn.1000-9604.2016.05.05
Abstract:
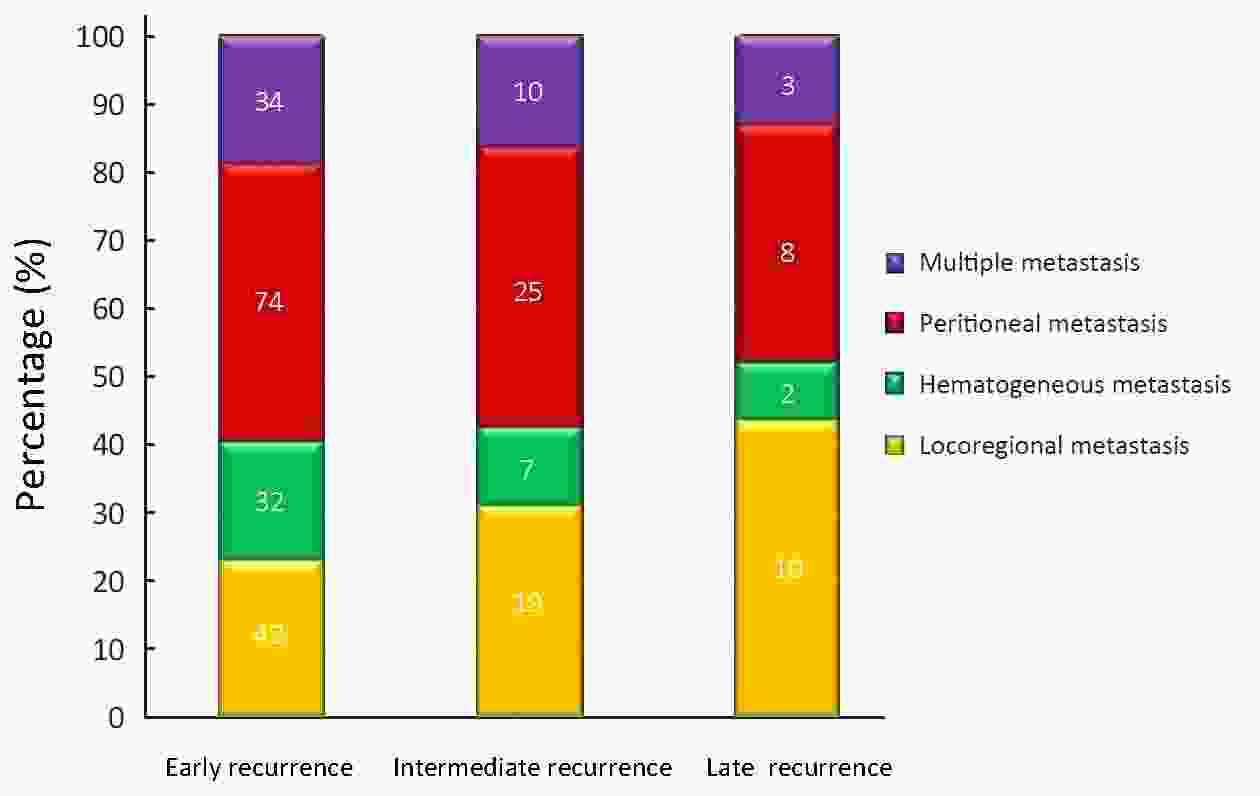
Objective: To investigate the characteristics of recurrences that occurred 5 or more years after curative resection for gastric cancer. Methods: We analyzed recurrences among 1,299 patients with gastric cancer who underwent curative operations at the Department of Surgery, Inje University Seoul Paik Hospital between September 1998 and December 2002. Recurrences were classified as within 2 years (early), 2–5 years (intermediate), and more than 5 years (late) after gastrectomy. The clinicopathologic findings of the patients with late recurrence were compared with those of patients in the other two recurrence groups, with special reference to the patterns of recurrence. Both univariate and multivariate analyses were performed, incorporating factors such as operation type, T-stage, N-stage, stage, lymphatic invasion, neural invasion, histology, tumor size, and recurrence site. Results: At the time of last follow-up, recurrence occurred in 266 (20.5%) patients. Recurrence times were classified as <2 years (182 patients), 2–5 years (61 patients), or >5 years (23 patients). The late recurrence rate was 8.6%. The occurrence of recurrence >5 years after gastrectomy was significantly correlated with age, operation type, T-stage, N-stage, stage, lymphatic invasion, neural invasion, histology, tumor size, location and recurrence site (P<0.05). The main recurrence patterns in the 23 patients with late recurrence were locoregional metastasis (10 patients, 43.5%), peritoneal seeding (8 patients, 34.8%), hematogenous metastasis (2 patients, 8.7%), and multiple metastasis (3 patients, 13.0%). A multivariate analysis showed that larger tumor size and younger age were independent prognostic factors for late recurrence. Additionally, locoregional and peritoneal recurrences were significantly more common than hematogenous recurrences. Conclusions: Although late recurrence was uncommon, younger age and larger tumor size were associated with high risk. Follow-up surveillance is recommended for locoregional and peritoneal metastasis.
2016, 28(5): 511-518.
doi: 10.21147/j.issn.1000-9604.2016.05.06
Abstract:
ObjectiveTo compare the numbers of positive and total lymph nodes and prognosis in gastric cancer patients whose perigastric lymph node retrieval was performed by surgeons and pathologists. MethodsWe conducted a retrospective analysis of clinical and follow-up data from 1, 056 patients who underwent gastric cancer D2 radical lymph node resection between January 2008 and December 2010 in the Gastrointestinal Surgery Department of Yantai Yuhuangding Hospital. The follow-up ended in December 2015. Patients were divided into two groups according to the specialty of physicians who performed the postoperative perigastric lymph node retrieval: the surgeon group (475 cases) and the pathologist group (581 cases). The numbers of positive and total perigastric lymph nodes and the 3- and 5-year survival were compared between gastric cancer patients in the two groups overall and stratified by TNM stage (the 7th Edition of the American Joint Committee on Cancer). ResultsOverall, the numbers of positive and total lymph nodes were significantly higher in the surgeon group than in the pathologist group (6.53±4.07 vs. 4.09±3.70, P=0.021; 29.64±11.50 vs. 20.71±8.56, P<0.001). Further analysis showed that the total number of lymph nodes in stage I patients (19.40±9.62 vs. 15.45±8.59, P=0.011) and the numbers of positive and total lymph nodes in stage II (1.38±1.08 vs. 0.87±1.55, P=0.031; 25.35±10.80 vs. 16.75±8.56, P<0.001) and stage III patients (8.11±6.91 vs. 6.66±5.12, P=0.026; 32.34±12.55 vs. 25.45±8.31, P<0.001) were significantly higher in the surgeon group than in the pathologist group. The survival analysis showed that the 3- and 5-year survival of stage II and III patients was significantly higher in the surgeon group than in the pathologist group (82.0% vs. 73.1%, 69.5% vs. 61.2%, P=0.038; 49.2% vs. 38.9%, 36.3% vs. 28.0%; P=0.045). ConclusionsCompared with retrieval performed by pathologists, postoperative perigastric lymph node retrieval performed by surgeons was associated with significant increase in the total lymph node number of stage I patients, the numbers of positive and total lymph nodes of stage II and III patients, and the survival of stage II and stage III gastric cancer patients.
2016, 20(5): 519-527.
doi: 10.21147/j.issn.1000-9604.2016.05.07
Abstract:
Objective To summarize experience in the diagnosis and treatment of pancreatic cystic neoplasms. Methods This is a retrospective study of 207 patients who were diagnosed with pancreatic cystic tumors at Peking Union Medical College Hospital between Jan 2009 and Mar 2014. Clinical data, such as clinical manifestations, radiological and pathological images and surgical recordings, were collected. Results Of the 207 included patients, females accounted for 76.81%, and the mean patient age was 52.04 years. Malignancy was more common in older patients who presented with marasmus and jaundice. Other risk factors included solid components in the tumor, a large tumor size, and elevated levels of tumor markers. Surgical treatment was required when a malignant tumor was suspected. The operation approach was selected based on the location, size and characteristics of the tumor. The position of the tumor relative to the pancreatic duct also played a significant role. Conclusions No specific symptoms were observed for the patients with pancreatic cystic tumors. Imaging played an important role in making a differential diagnosis. Furthermore, surgical treatment should be proposed for patients with significant symptoms and potentially malignant tumors. The tumor resection rate is high, suggestive of good prognosis.
Simultaneous hepatic resection benefits patients with synchronous colorectal cancer liver metastases
2016, 20(5): 528-535.
doi: 10.21147/j.issn.1000-9604.2016.05.08
Abstract:
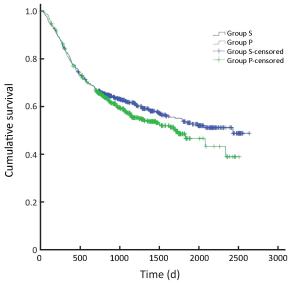
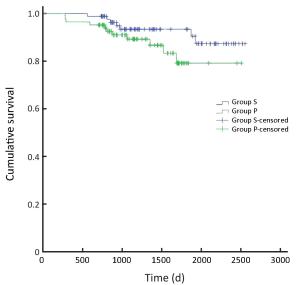
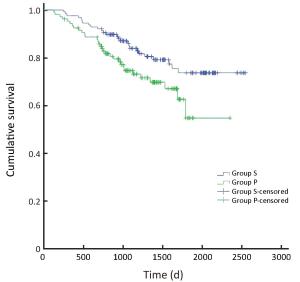
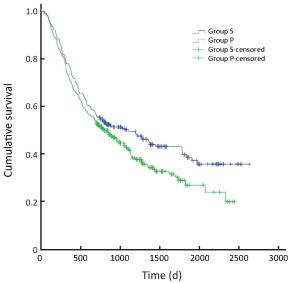
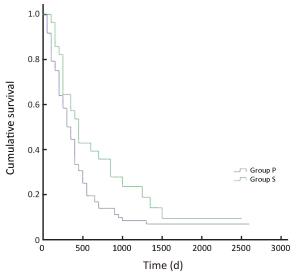
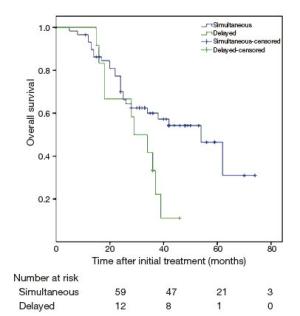
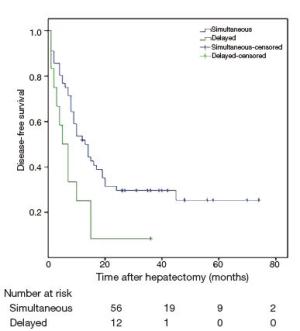
Objective The safety of the simultaneous resection of synchronous colorectal cancer liver metastases (SCRLM) is still being debated. However, this simultaneous operative approach is more commonly adopted at present than in the past. Therefore, we compared simultaneous hepatic resection with delayed hepatic resection in this study. Methods All patients with SCRLM diagnosed before initial treatment between January 2009 and September 2013 were retrospectively included in our study. Short-term and long-term outcomes were compared in patients who underwent simultaneous colorectal and hepatic resection and those treated by delayed hepatectomy. Results Among the 73 patients diagnosed with SCRLM, simultaneous colorectal and hepatic resection was performed in 60 patients (82.2%), while delayed hepatic resection was performed in 13 patients (17.8%). The mortality rate was zero. The postoperative complication rate after delayed resection was higher than, but not significantly different from, that after simultaneous resection (46% vs. 23%, P=0.166). The duration of operating time (240 vs. 420 min, P<0.05) and postoperative hospital stay time (11 vs. 18 days, P<0.05) were shorter in the simultaneous resection group. After the initial treatments were given, the 1-, 2-, and 3-year survival rates in the simultaneous resection group were 77%, 59%, and 53%, respectively, whereas those in the delayed resection group were 67%, 42%, and 10%, respectively. The 5-year survival rate in the simultaneous resection group was 23%; overall survival differed significantly between the two groups (P=0.037). Median disease-free survival (DFS) times were 19.1 months in the simultaneous resection group and 8.8 months in the delayed resection group. DFS differed significantly between the two groups. Conclusions Simultaneous colorectal and hepatic resection is safe and exhibits advantages in the long-time survival of patients.
2016, 28(5): 536-542.
doi: 10.21147/j.issn.1000-9604.2016.05.09
Abstract:
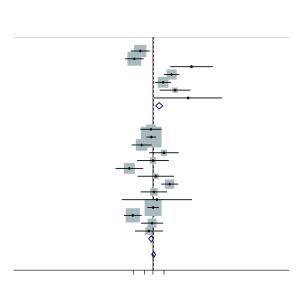
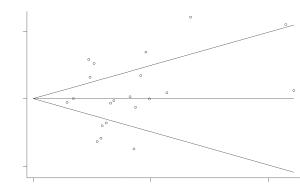
Objective Many studies have examined the association between the TNF-308 G/A polymorphism gene polymorphisms and hepatocellular carcinoma risk in various populations, but their results have been inconsistent. To assess this relationship more precisely, a meta-analysis was performed. Methods The PubMed and CNKI (China National Knowledge Infrastructure) database was searched for case-control studies. Odds ratios (OR) with 95% CIs were used to determine the strength of association between the TNF-308 G/A polymorphisms and HCC risk. The pooled ORs for the risk associated with the TNF-308 G/A genotype, the A carriers (A/G + A/A) vs. the wild-type homozygotes (G/G), A/A vs. G/G were calculated, respectively. Subgroup analyses were done by ethnicity and smoking status. Heterogeneity assumptions were assessed by chi-square-based Q-test. Results Ultimately, 21 studies, comprising 2,923 hepatocellular carcinoma cases and 4,323 controls were included. Overall, the A carriers (G/A + A/A) vs. the wild-type homozygotes (G/G), the pooled OR was 1.05 (95% CI, 0.93-1.19; P=0.000 for heterogeneity), for A/A vs. G/G the pooled OR was 1.07 (95% CI, 0.95-1.21; P=0.007 for heterogeneity). In the stratified analysis by ethnicity, the significantly risks were found among non-Asians. However, for Asians, significantly risks were not found. Conclusions The TNF-308 G/A polymorphisms are not associated with hepatocellular carcinoma risk among Asians, but for non-Asians.
2016, 28(5): 543-552.
doi: 10.21147/j.issn.1000-9604.2016.05.10
Abstract:
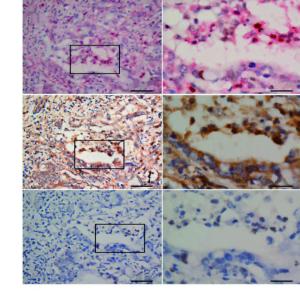
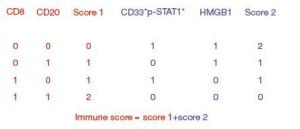
Objective There is heterogeneity in the prognosis of gastric cancers staged according to the tumornodes- metastasis (TNM) system. This study evaluated the prognostic potential of an immune score system to supplement the TNM staging system. Methods An immunohistochemical analysis was conducted to assess the density of T cells, B cells, and myeloid-derived suppressor cells (MDSCs) in cancer tissues from 100 stage IIIA gastric cancer patients; the expression of the high-mobility group protein B1 (HMGB1) was also evaluated in cancer cells. The relationship between the overall survival (OS), disease-free survival (DFS), and immunological parameters was analyzed. Results An immune score system was compiled based on the prognostic role of the density of T cells, B cells, MDSCs, and the expression of HMGB1 in cancer tissues. The median 5-year survival of this group of patient was 32%. However, the 5-year survival rates of 80.0%, 51.7%, 0%, 5.8%, and 0% varied among the patients with an immune score of 4 to those with an immune score of 0 based on the immune score system, respectively. Similarly, differences in DFS rates were observed among the immune score subgroups. Conclusions An immune score system could effectively identify the prognostic heterogeneity within stage IIIA gastric cancer patients, implying that this immune score system may potentially supplement the TNM staging system, and help in identifying a more homogeneous group of patients who on the basis of prognosis can undergo adjuvant therapy.

 Abstract
Abstract FullText HTML
FullText HTML PDF 619KB
PDF 619KB