2018 Vol.30(2)
Display Mode: |
2018, 30(2): 157-172.
doi: 10.21147/j.issn.1000-9604.2018.02.01
Abstract:
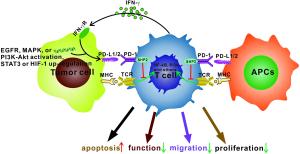
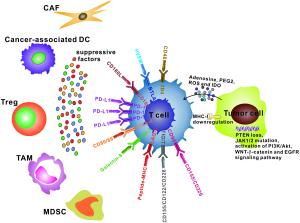
Programmed cell death 1 (PD-1)/programmed cell death 1 ligand (PD-L1) blockade has shown promising effects in cancer immunotherapy. Removing the so-called " brakes” on T cell immune responses by blocking the PD-1/PD-L1 check point should boost anti-tumor immunity and provide durable tumor regression for cancer patients. However, 30%–60% of patients show no response to PD-1/PD-L1 blockade. Thus, it is urgent to explore the underlying resistance mechanisms to improve sensitivity to anti-PD-1/PD-L1 therapy. We propose that the mechanisms promoting resistance mainly include T cell exclusion or exhaustion at the tumor site, immunosuppressive factors in the tumor microenvironment (TME), and a range of tumor-intrinsic factors. This review highlights the power of studying the cellular and molecular mechanisms of resistance to improve the rational design of combination therapeutic strategies that can be translated to the clinic. Here, we briefly discuss the development of PD-1/PD-L1 blockade agents and focus on the current issues and future prospects for potential combinatorial therapeutic strategies that include anti-PD-1/PD-L1 therapy, based upon the available preclinical and clinical data.
Programmed cell death 1 (PD-1)/programmed cell death 1 ligand (PD-L1) blockade has shown promising effects in cancer immunotherapy. Removing the so-called " brakes” on T cell immune responses by blocking the PD-1/PD-L1 check point should boost anti-tumor immunity and provide durable tumor regression for cancer patients. However, 30%–60% of patients show no response to PD-1/PD-L1 blockade. Thus, it is urgent to explore the underlying resistance mechanisms to improve sensitivity to anti-PD-1/PD-L1 therapy. We propose that the mechanisms promoting resistance mainly include T cell exclusion or exhaustion at the tumor site, immunosuppressive factors in the tumor microenvironment (TME), and a range of tumor-intrinsic factors. This review highlights the power of studying the cellular and molecular mechanisms of resistance to improve the rational design of combination therapeutic strategies that can be translated to the clinic. Here, we briefly discuss the development of PD-1/PD-L1 blockade agents and focus on the current issues and future prospects for potential combinatorial therapeutic strategies that include anti-PD-1/PD-L1 therapy, based upon the available preclinical and clinical data.
2018, 30(2): 173-196.
doi: 10.21147/j.issn.1000-9604.2018.02.02
Abstract:
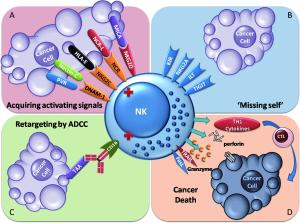
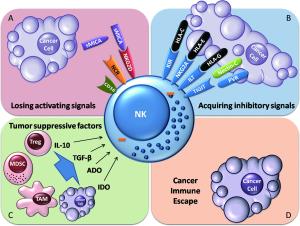
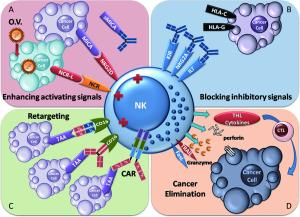
A group of impressive immunotherapies for cancer treatment, including immune checkpoint-blocking antibodies, gene therapy and immune cell adoptive cellular immunotherapy, have been established, providing new weapons to fight cancer. Natural killer (NK) cells are a component of the first line of defense against tumors and virus infections. Studies have shown dysfunctional NK cells in patients with cancer. Thus, restoring NK cell antitumor functionality could be a promising therapeutic strategy. NK cells that are activated and expanded ex vivo can supplement malfunctional NK cells in tumor patients. Therapeutic antibodies, chimeric antigen receptor (CAR), or bispecific proteins can all retarget NK cells precisely to tumor cells. Therapeutic antibody blockade of the immune checkpoints of NK cells has been suggested to overcome the immunosuppressive signals delivered to NK cells. Oncolytic virotherapy provokes antitumor activity of NK cells by triggering antiviral immune responses. Herein, we review the current immunotherapeutic approaches employed to restore NK cell antitumor functionality for the treatment of cancer.
A group of impressive immunotherapies for cancer treatment, including immune checkpoint-blocking antibodies, gene therapy and immune cell adoptive cellular immunotherapy, have been established, providing new weapons to fight cancer. Natural killer (NK) cells are a component of the first line of defense against tumors and virus infections. Studies have shown dysfunctional NK cells in patients with cancer. Thus, restoring NK cell antitumor functionality could be a promising therapeutic strategy. NK cells that are activated and expanded ex vivo can supplement malfunctional NK cells in tumor patients. Therapeutic antibodies, chimeric antigen receptor (CAR), or bispecific proteins can all retarget NK cells precisely to tumor cells. Therapeutic antibody blockade of the immune checkpoints of NK cells has been suggested to overcome the immunosuppressive signals delivered to NK cells. Oncolytic virotherapy provokes antitumor activity of NK cells by triggering antiviral immune responses. Herein, we review the current immunotherapeutic approaches employed to restore NK cell antitumor functionality for the treatment of cancer.
2018, 30(2): 209-221.
doi: 10.21147/j.issn.1000-9604.2018.02.04
Abstract:
Two major treatment modalities in cervical cancer are radiation therapy (RT) and surgery. Chemotherapy continues to be the main form of systemic therapy adjunctive to definitive local therapies, and is used for palliation. Platinum-based regimens, administered concurrently with both definitive and postoperative RT, were demonstrated to provide significant survival benefits, whereas the beneficial effect of concurrent chemoradiotherapy in later-stage disease was smaller. The role of chemotherapy in addition to RT in IB1/IIA1 cervical cancer patients not undergoing surgery remains undefined. Likewise, the role of chemotherapy in combination with postoperative RT for patients with intermediate-risk factors for recurrence has not yet been verified. The recent standard for chemoradiotherapy is cisplatin alone administered weekly. Other cisplatin-based or non-cisplatin-based regimens have not been subjected to large clinical studies. The benefits of consolidation chemotherapy after chemoradiation for locally advanced cervical cancer are still undetermined. Neoadjuvant cisplatin-based chemotherapy followed by surgery has shown survival benefits, however its role in the era of chemoradiotherapy remains unclear. The combination of cisplatin and paclitaxel is considered a standard regimen in the palliative setting. There is no standard of care for second-line systemic therapy in advanced cervical cancer. Bevacizumab combined with palliative chemotherapy (cisplatin/paclitaxel or topotecan/paclitaxel) in the first-line treatment for recurrent/metastatic cervical cancer significantly improves overall survival when compared to chemotherapy alone. The role of immunotherapy in cervical cancer remains to be established. The optimal combined modality treatment including systemic therapy for cervical tumors of non-squamous histology remains a matter of debate. Ongoing accumulation of data on genomic and proteomic characteristics provides insight into the molecular heterogeneity of cervical cancer and paves the way for developing molecularly targeted therapies.
Two major treatment modalities in cervical cancer are radiation therapy (RT) and surgery. Chemotherapy continues to be the main form of systemic therapy adjunctive to definitive local therapies, and is used for palliation. Platinum-based regimens, administered concurrently with both definitive and postoperative RT, were demonstrated to provide significant survival benefits, whereas the beneficial effect of concurrent chemoradiotherapy in later-stage disease was smaller. The role of chemotherapy in addition to RT in IB1/IIA1 cervical cancer patients not undergoing surgery remains undefined. Likewise, the role of chemotherapy in combination with postoperative RT for patients with intermediate-risk factors for recurrence has not yet been verified. The recent standard for chemoradiotherapy is cisplatin alone administered weekly. Other cisplatin-based or non-cisplatin-based regimens have not been subjected to large clinical studies. The benefits of consolidation chemotherapy after chemoradiation for locally advanced cervical cancer are still undetermined. Neoadjuvant cisplatin-based chemotherapy followed by surgery has shown survival benefits, however its role in the era of chemoradiotherapy remains unclear. The combination of cisplatin and paclitaxel is considered a standard regimen in the palliative setting. There is no standard of care for second-line systemic therapy in advanced cervical cancer. Bevacizumab combined with palliative chemotherapy (cisplatin/paclitaxel or topotecan/paclitaxel) in the first-line treatment for recurrent/metastatic cervical cancer significantly improves overall survival when compared to chemotherapy alone. The role of immunotherapy in cervical cancer remains to be established. The optimal combined modality treatment including systemic therapy for cervical tumors of non-squamous histology remains a matter of debate. Ongoing accumulation of data on genomic and proteomic characteristics provides insight into the molecular heterogeneity of cervical cancer and paves the way for developing molecularly targeted therapies.
2018, 30(2): 197-208.
doi: 10.21147/j.issn.1000-9604.2018.02.03
Abstract:
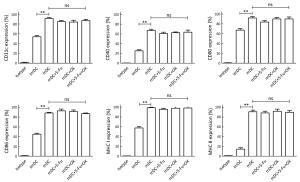
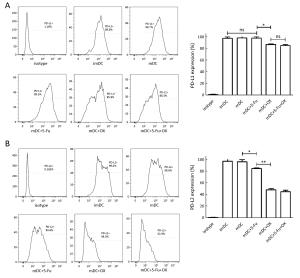
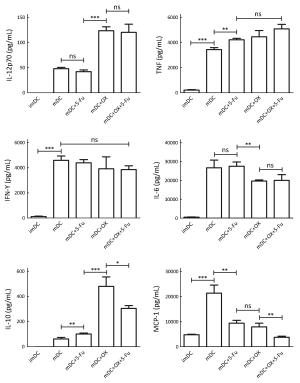
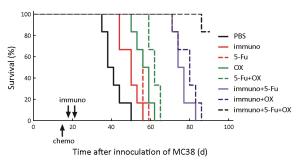
Objective The aim of the present study was to investigate the effects of 5-fluorouracil (5-Fu) and oxaliplatin on the function and activation pathways of mouse dendritic cells (DCs), and to clarify whether 5-Fu/oxaliplatin combined with the CD1d-MC38/α-galactosylceramide (α-GC) tumor vaccine exhibits synergistic effects on the treatment of colon cancer in mice. Methods The combination of the Toll like receptor (TLR) ligands and/or 5-Fu/oxaliplatin was added into myeloid-derived DCs in vitro culture. DC phenotypic changes were detected by flow cytometry, and the secretion of DC cytokines was detected by cytometric bead array (CBA). A MC38 mouse colon cancer model was constructed and the DCs were isolated from the spleen, tumor tissue and lymph nodes following intraperitoneal injection of 5-Fu/oxaliplatin. The cell phenotypes were detected by flow cytometry. The tumor infiltrating leukocytes, splenocytes and lymph node cells were co-cultured with the dead MC38 tumor cells, and the secretion levels of interferon-γ (IFN-γ) were detected. 5-Fu/oxaliplatin combined with our previously developed CD1d-MC38/α-GC tumor vaccine was used to inhibit the growth of MC38 colon cancer in mice, and the tumor growth rate and survival time were recorded. Results 5-Fu/oxaliplatin exerted no significant effect on the expression of the stimulating phenotypes of DCs in vitro, while it could reduce the expression of programmed death ligand 1/2 (PD-L1/L2) and promote interleukin-12 (IL-12) secretion by DCs. Furthermore 5-Fu/oxaliplatin was beneficial to the differentiation of T-helper 1 (Th1) cells. 5-Fu/oxaliplatin further enhanced the stimulating phenotypic expression of DCs in tumor bearing mice, decreased PD-L1/L2 expression, and specifically activated the lymphocytes. The CD1d-MC38/α-GC tumor vaccine combined with 5-Fu/oxaliplatin could exert a synergistic role that resulted in a significant delay of the tumor growth rate, and an increase in the survival time of tumor bearing mice. Conclusions 5-Fu/oxaliplatin decreased the expression of the DC inhibitory phenotypes PD-L1/L2, promoted DC phenotypic maturation in tumor bearing mice, activated the lymphocytes of tumor bearing mice, and exerted synergistic effects with the CD1d-MC38/α-GC colon cancer tumor vaccine.
2018, 30(2): 222-230.
doi: 10.21147/j.issn.1000-9604.2018.02.05
Abstract:
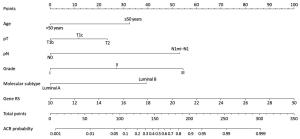
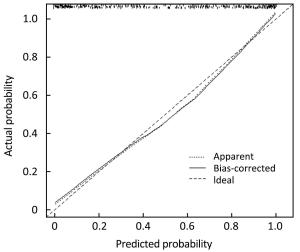
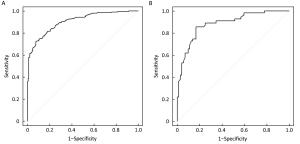
Objective The indication of adjuvant chemotherapy recommendation (ACR) in breast cancer patients with intermediate recurrence score (RS) is controversial. This study investigated the relationship between routine clinicopathological indicators and ACR, and established a nomogram for predicting the probability of ACR in this subset of patients. Methods Data for a total of 504 consecutive patients with intermediate RS from January 2014 to December 2016 were retrospectively reviewed. A nomogram was constructed using a multivariate logistic regression model based on data from a training set (378 cases) and validated in an independent validation set (126 cases). A Youden-derived cut-off value was assigned to the nomogram for accuracy evaluation. Results The multivariate logistic regression analysis identified that age, histological grade, tumor size, lymph node (LN) status, molecular subtype, and RS were independent predictors of ACR. A nomogram based on these predictors performed well. The P value of the Hosmer-Lemeshow test for the prediction model was 0.286. The area under the curve (AUC) values were 0.905 [95% confidence interval (95% CI): 0.876–0.934] and 0.883 (95% CI: 0.824–0.942) in the training and validation sets, respectively. The accuracies of the nomogram for ACR were 84.4% in the training set and 82.1% in the validation set. Conclusions We developed a nomogram to predict the probability of ACR in breast cancer patients with intermediate RS. This model may aid the individual risk assessment and guide treatment decisions in clinical practice.
2018, 30(2): 231-239.
doi: 10.21147/j.issn.1000-9604.2018.02.06
Abstract:
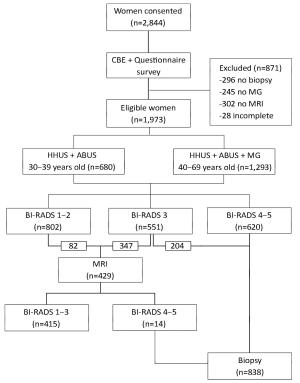
Objective The automated breast ultrasound system (ABUS) is a potential method for breast cancer detection; however, its diagnostic performance remains unclear. We conducted a hospital-based multicenter diagnostic study to evaluate the clinical performance of the ABUS for breast cancer detection by comparing it to handheld ultrasound (HHUS) and mammography (MG). Methods Eligible participants underwent HHUS and ABUS testing; women aged 40–69 years additionally underwent MG. Images were interpreted using the Breast Imaging Reporting and Data System (BI-RADS). Women in the BI-RADS categories 1–2 were considered negative. Women classified as BI-RADS 3 underwent magnetic resonance imaging to distinguish true- and false-negative results. Core aspiration or surgical biopsy was performed in women classified as BI-RADS 4–5, followed by a pathological diagnosis. Kappa values and agreement rates were calculated between ABUS, HHUS and MG. Results A total of 1,973 women were included in the final analysis. Of these, 1,353 (68.6%) and 620 (31.4%) were classified as BI-RADS categories 1–3 and 4–5, respectively. In the older age group, the agreement rate and Kappa value between the ABUS and HHUS were 94.0% and 0.860 (P<0.001), respectively; they were 89.2% and 0.735 (P<0.001) between the ABUS and MG, respectively. Regarding consistency between imaging and pathology results, 78.6% of women classified as BI-RADS 4–5 based on the ABUS were diagnosed with precancerous lesions or cancer; which was 7.2% higher than that of women based on HHUS. For BI-RADS 1–2, the false-negative rates of the ABUS and HHUS were almost identical and were much lower than those of MG. Conclusions We observed a good diagnostic reliability for the ABUS. Considering its performance for breast cancer detection in women with high-density breasts and its lower operator dependence, the ABUS is a promising option for breast cancer detection in China.
2018, 30(2): 240-253.
doi: 10.21147/j.issn.1000-9604.2018.02.07
Abstract:
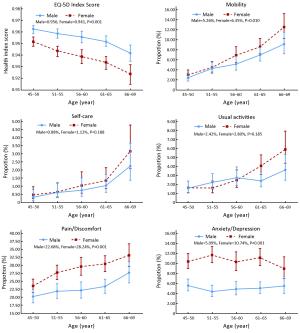
Objective There have been few population-based studies evaluating health related quality of life (HRQOL) in rural populations in China, and this study aimed to assess the current status of and risk factors for HRQOL in a general rural population in high risk region of esophageal cancer in China. Methods From November 2015 to September 2016, 12,085 permanent residents aged 45–69 years from 257 villages in the Endoscopic Screening for Esophageal Cancer in China (ESECC) trial (ClinicalTrials.gov: NCT01688908) randomly selected from Hua County, Henan Province, China were interviewed. The EQ-5D-3L, a generic measure of HRQOL, and a questionnaire were used to assess their HRQOL and potential risk factors. Results Among all the participants, 30.62% of the participants reported problems in at least one EQ-5D dimension. Pain/discomfort (25.52%) was the most frequently reported problem followed by anxiety/depression (7.97%), mobility (5.82%), usual activities (2.61%) and self-care (1%). These rural residents had a mean EQ-5D index score of 0.948, and lower EQ-5D index scores were associated with older age, female gender, lower levels of household annual per capita income, living alone, using shallow wells as main source of drinking water, exposure to family members smoking, testiness, unhealthy dietary habits, overweight or obesity, upper gastrointestinal cancer related symptoms and chronic diseases. Conclusions Rural residents in China have a relatively low quality of life. Health promotion programs in this population should focus on the elderly, especially elderly women and the elderly living alone. Improving basic living circumstances and primary medical care services should be priorities. Results of this study will also serve as the basis for the cost-utility evaluation in our ESECC screening trial.
2018, 30(2): 254-262.
doi: 10.21147/j.issn.1000-9604.2018.02.08
Abstract:
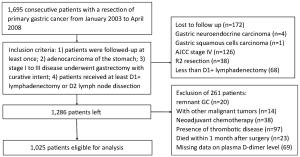
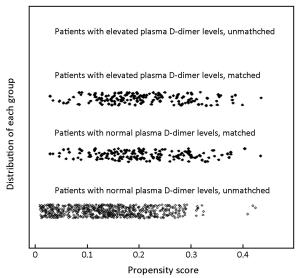
Objective Elevated plasma D-dimer has been reported to be associated with advanced tumor stage and poor survival in several types of malignancies. The purpose of this study was to assess the potential impact of preoperative plasma D-dimer level (PDL) on overall survival (OS) of gastric cancer (GC) patients undergoing curative surgery by applying propensity score analysis. Methods A total of 1,025 curatively resected GC patients in Tianjin Medical University Cancer Institute & Hospital were enrolled. Patients were categorized into two groups based on preoperative PDL: the elevated group (EG) and the normal group (NG). To overcome bias due to the different distribution of covariates for the two groups, a one-to-one match was applied using propensity score analysis, after matching, prognostic factors were analyzed. Results In analysis for the whole study series, patients in the EG were more likely to have a larger proportion of tumor size ≥5 cm (67.5% vs. 55.8%, P=0.006), elder mean age (64.0±10.8 years vs. 60.5±11.6 years, P<0.001) and advanced tumor (T), node (N), and TNM stage. Patients with elevated PDL demonstrated a significantly lower 5-year OS than those with normal PDL (27.0% vs. 42.6%, P<0.001), however, the PDL was not an independent prognostic factor for OS in multivariate analysis [hazard ratio: 1.13, 95% confidence interval (95% CI): 0.92–1.39, P=0.236]. After matching, 163 patients in the EG and 163 patients in the NG had the same characteristics. The 5-year OS rate for patients in the EG was 27.0% compared with 25.8% for those in the NG (P=0.809, log-rank). Conclusions The poor prognosis of GC patients with elevated preoperative PDL was due to the advanced tumor stage and elder age rather than the elevated D-dimer itself.
2018, 30(2): 263-271.
doi: 10.21147/j.issn.1000-9604.2018.02.09
Abstract:
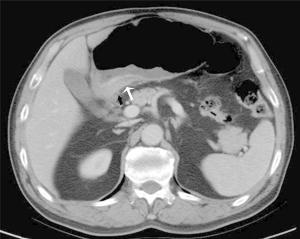
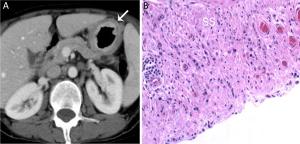
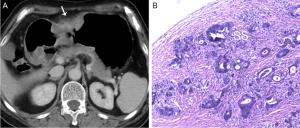
Objective To study the value of high enhanced serosa sign on contrast-enhanced computed tomography (CT) in differentiating T3 from T4a gastric cancer in different Lauren classification. Methods This study included 276 consecutive patients with surgically confirmed pT3 or pT4a gastric cancers. The pre-operative CT images were reviewed by two radiologists blinded. The demonstration of the high enhanced serosa on CT between T3 and T4a was compared with chi-square test. The diagnostic performance of this sign on CT in the differentiation of T4a from T3 in different Lauren classification was calculated. Results The accuracy, sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) for the judgement of serosa invasion using the high enhanced serosa sign on CT was 74.6%, 63.7%, 83.6%, 76.0% and 73.8% by one radiologist and 76.4%, 66.1%, 84.9%, 78.1% and 75.4% by the other radiologist. Compared to the intestinal-type, the sensitivity of the judgement of serosa invasion using the high enhanced serosa sign on CT in diffuse-type was significant higher (80% in both readers), while the specificity trended to be lower (65.9% and 80.5%, respectively). There is no significant difference in the accuracy of diagnosis between intestinal-type and diffuse-type of gastric cancers (the P-values of two radiologists were 0.968, 0.591, respectively). The combination of the high enhanced serosa sign with conventional CT signs is significant different in diagnosis of T3 and T4a (P<0.001). The diagnostic accuracy was increased in both radiologists after the combination. The two readers achieved substantial agreement, with Kappa coefficient of 0.63, P<0.001. Conclusions The high enhanced serosa sign on CT is associated with serosa involvement. The sensitivity of the judgement of serosa invasion using this sign on CT in diffuse-type was significant higher than that in intestinal-type.
2018, 30(2): 272-281.
doi: 10.21147/j.issn.1000-9604.2018.02.10
Abstract:
Objective For patients with locally advanced rectal cancer treated with neoadjuvant chemoradiotherapy (NCRT), significant pathological response of the primary tumor has been proposed to identify candidates for organ preservation. However, this does not address metastatic lymph nodes in the mesorectum. The aim of this study was to assess the incidence of lymph node metastases in ypT0 patients treated with NCRT and curative resection and to explore risk factors associated with survival. Methods This was a retrospective study of patients with ypT0 rectal cancer after NCRT and curative resection at a tertiary care center in China from 2005 to 2014. Results A total of 60 (18.6%) patients who underwent surgery after NCRT and achieved ypT0 were enrolled in this study; one patient was excluded owing to lack of follow-up. Of these 59 patients, lymph node metastases were found in the mesorectum (ypT0N+) in eight (13.6%) patients. After a median follow-up of 52 months, 5-year recurrence-free survival (82.7% vs. 62.5%, P=0.014) and overall survival (OS) (90.9% vs. 70.0%, P=0.032) were much higher in ypN0 than ypN+ patients. Multivariate analyses showed that ypN+ status (P=0.009) and perioperative blood transfusion (BT) (P=0.001) were significantly independent risk factors associated with recurrence; however, no factor was correlated with 5-year OS. Conclusions Patients with ypT0N0 rectal cancer can achieve excellent long-term outcomes; however, positive lymph nodes or tumor deposits can still be found in 13.6% of ypT0 patients. Nodal positivity in the mesorectum and perioperative BT are independent risk factors for recurrence.
2018, 30(2): 282-290.
doi: 10.21147/j.issn.1000-9604.2018.02.11
Abstract:
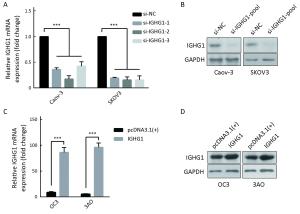
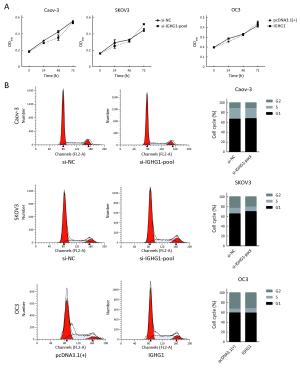
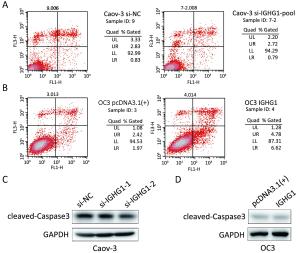
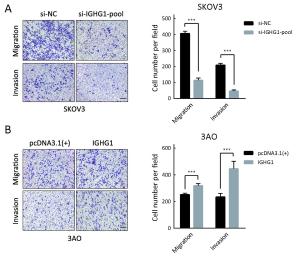
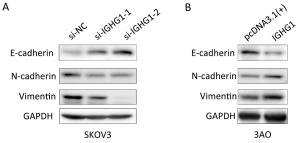
Objective Ovarian cancer (OC) is one of the leading causes of death for female cancer patients. COC166-9 is an OC-specific monoclonal antibody and we have identified immunoglobulin γ-1 heavy chain constant region (IGHG1) as its antigen. We explore the function of IGHG1 in proliferation, apoptosis and motility of OC cells further in this research. Methods IGHG1 expression in OC specimens was detected through immunohistochemistry. Real-time quantitative polymerase chain reaction (RT-qPCR) or western blotting assay was used to test IGHG1 expression in OC cells. Viability of OC cells was tested by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Flow cytometry or western blotting assay was used to detect cell cycle and apoptosis. Cellular motility was analyzed by using transwell assay and the markers of epithelial-mesenchymal transition (EMT) were tested through immunoblots. Results Although it exerts negligible effect on the viability and apoptosis of OC cells, IGHG1 could promote migration and invasion of malignant cells in vitro. Mechanistically, IGHG1 increases the expression of N-cadherin and Vimentin while decreases E-cadherin expression. Additionally, IGHG1 expression in OC specimens is higher relative to the paired normal counterparts. Further analysis demonstrates that the increased IGHG1 expression correlates positively with the lymph node metastasis of OC. Conclusions IGHG1 promotes the motility of OC cells likely through executing the EMT program. Increased IGHG1 expression in OC specimens is associated with the lymph node metastasis.

 Abstract
Abstract FullText HTML
FullText HTML PDF 1100KB
PDF 1100KB